|
The term and the concept are used in diverse fields, from classical thermodynamics, where it was first recognized, to the microscopic description of nature in statistical. This concept was introduced by a German physicist named Rudolf Clausius in the year 1850. Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. The higher the entropy of an object, the more uncertain we are about the states. Generally, entropy is defined as a measure of randomness or disorder of a system. Entropy is a thermodynamic quantity that is generally used to describe the course of a process, that is, whether it is a spontaneous process and has a probability of occurring in a defined direction, or a non-spontaneous process and will not proceed in the defined direction, but in the reverse direction. In this sense, entropy is a measure of uncertainty or randomness. Entropy is also a measure of the number of possible arrangements the atoms in a system can have. However, the entropic quantity we have defined is very useful in defining whether a given reaction will occur. The entropy of an object is a measure of the amount of energy which is unavailable to do work. 
Newman b : a process of degradation or running down or a trend to disorder The deterioration of copy editing and proof-reading, incidentally, is a token of the cultural entropy that has overtaken us in the postwar years. 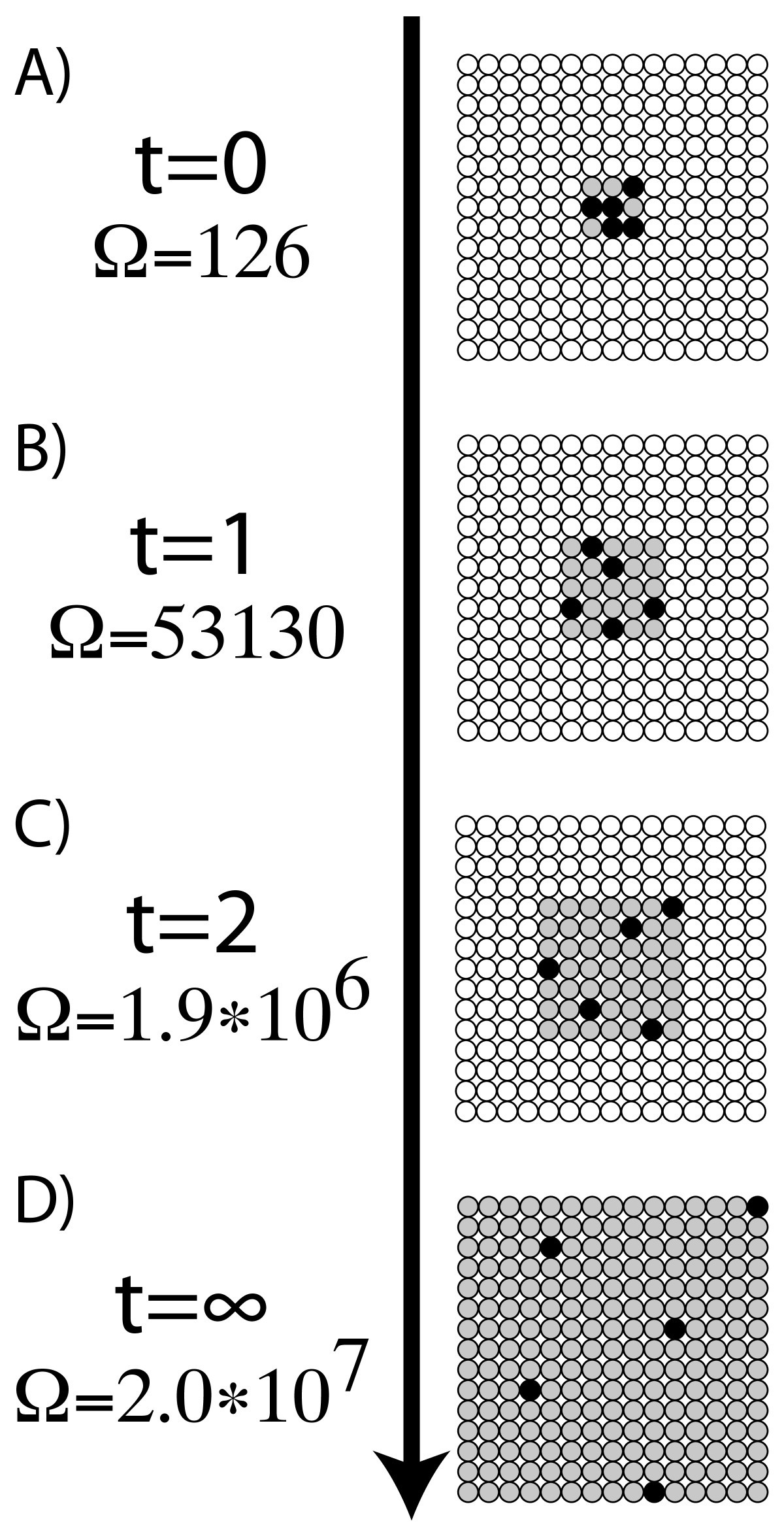
We have a closed system if no energy from an outside source can enter the system. ENTROPY English meaning - Cambridge Dictionary Meaning of entropy in English entropy noun U specialized uk / en.tr.pi / us / en.tr. Entropy is the general trend of the universe toward death and disorder. Unfortunately, in the information theory, the symbol for entropy is Hand the constant k B is absent. In science, entropy is used to determine the amount of disorder in a closed system. This expression is called Shannon Entropy or Information Entropy. It is evident from our experience that ice melts, iron rusts, and gases mix together. Entropy is a measure of the amount of energy that is unavailable to do work in a closed system. This apparent discrepancy in the entropy change between an irreversible and a reversible process becomes clear when considering the changes in entropy of the surrounding and system, as described in the second law of thermodynamics.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
